Garbh-Ini
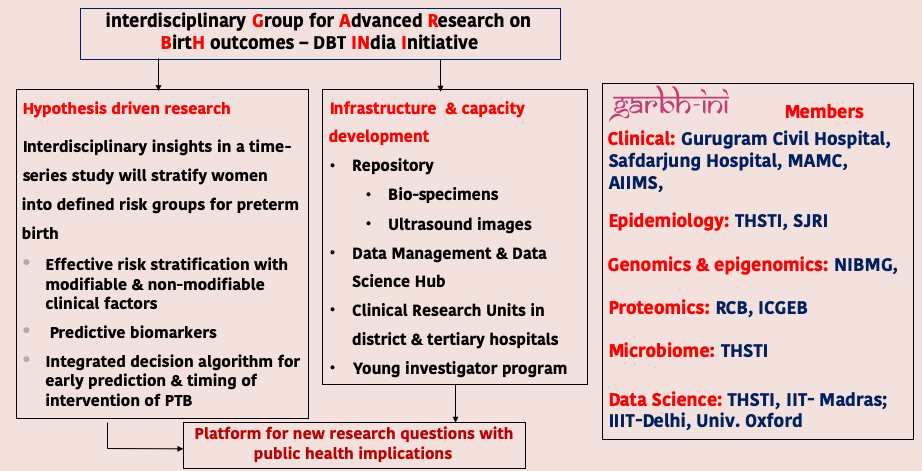
interdisciplinary Group for Advanced research on BirtH outcomes -DBT INdia Initiative
interdisciplinary Group for Advanced research on BirtH outcomes -DBT INdia Initiative
Garbh-ini (Interdisciplinary Group for Advanced Research on Birth Outcomes—DBT India Initiative) was initiated by the Department of Biotechnology (DBT), in 2014 as a collaborative interdisciplinary program. This program is led by Translational Health Science and Technology Institute (THSTI), NCR Biotech cluster, Faridabad.
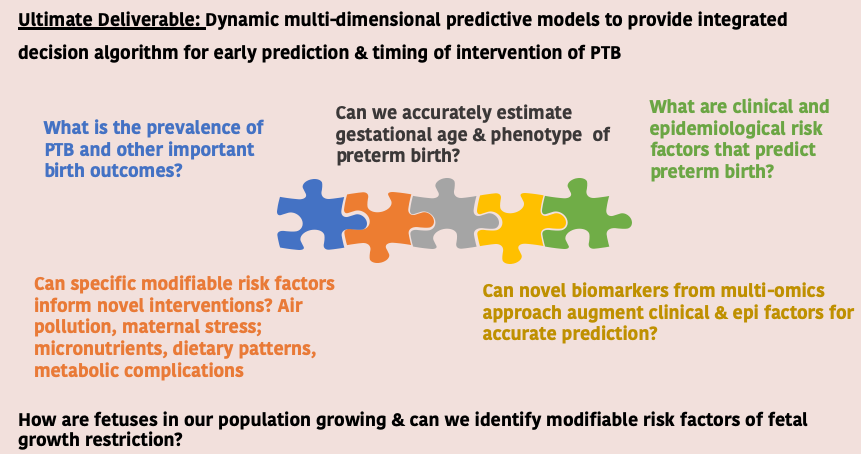
Garbh-Ini is a cohort study of pregnant women initiated in May 2015 at the civil hospital in Gurugram, Haryana, India. Women are enrolled within 20 weeks of gestation and are followed until delivery and once postpartum. The objectives are to identify clinical, epidemiologic, genomic, epigenomic, proteomic, and microbial correlates; discover molecular-risk markers by using an integrative -omics approach; and generate a risk-prediction algorithm for preterm birth. Three DBT institutes and the Gurugram civil hospital, Government of Haryana have partnered with each other to make this initiative possible Read more…
Globally, preterm birth is a major public health problem. In India, 3.6 million of the 27 million babies born annually are preterm. Risk stratification of women based on multidimensional risk factors assessed during pregnancy is critical for prevention of preterm birth. An interdisciplinary Group for Advanced Research in BirtH outcomes- DBT India Initiative (GARBH-Ini) has initiated a cohort study of pregnant women has been initiated in May 2015 at the civil hospital in Gurugram, Haryana, India with the ultimate objective (i) to acquire deep fundamental knowledge on adverse pregnancy outcomes such as preterm birth (PTB) and fetal growth restriction (FGR) and (ii) apply this knowledge to identify efficient and sustainable solutions that would assist in reducing associated mortality, immediate and long term morbidity.
Accounting for a cohort design to detect epidemiological risk factors & nested case-control design for identifying biomarkers, an apriori sample size of 8000 was estimated.
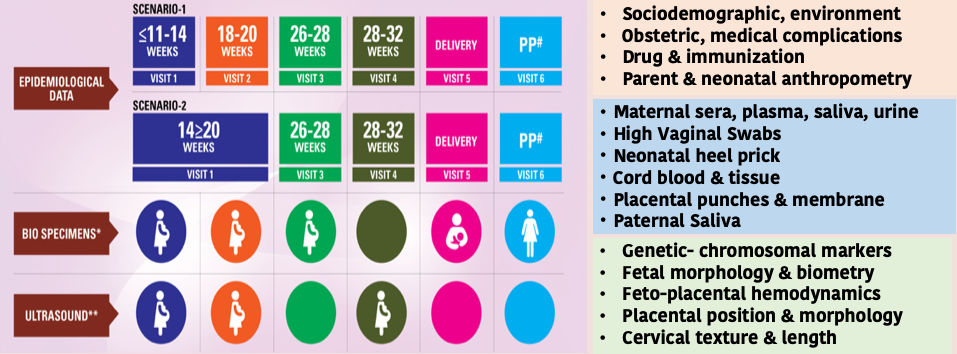
We are enrolling pregnant women early in pregnancy and certainly before <20 weeks of gestation and willing to continue their care till delivery at GCH.
The enrolled women are followed up 4-5 time-points across 3 trimesters of pregnancy to document extensive clinical & epidemiological information, varied maternal and neonatal biospecimens and perform serial ultrasonographic examination.